Advances in pain control in palliative care
Pain in palliative care needs to be carefully assessed before treatment.
RENE KRAUSE, MB ChB, MFamMed, MPhil (Palliative Medicine)
Family Physician, St Luke’s Hospice, Cape Town
Rene Krause has been working in palliative care for the past 7
years. She worked in Bethesda Hospice and in Harry Comay TB hospital
with special interest in MDR TB. For the past 3 years she has been
working at St Luke’s Hospice, Kenilworth, Cape Town, where she is
the doctor in a 10-bed IPU which mainly concentrates on cancer patients
and pain control.
JANET STANFORD, MB ChB, MPhil (Maternal and Child Health), MPhil (Palliative Medicine)
Medical Officer and CEO, Knysna Sedgefield Hospice
Janet Stanford is married to John Stanwix, economist turned farmer.
She has 3 children Emily, Ben and Louisa and enjoys beekeeping and yoga.
Corresponding author: Rene Krause (renek@stlukes.co.za)
The recent PainSA conference held in conjunction with the
Inter-national Association of the study of Pain, Africa Association for
the study of Pain, NeuroPsig and the London Pain Consortium, presented
current physiological evidence of the importance of neuronal and
synaptic activities involved in generating pain. The physiological
evidence supports the inclusion of medication previously considered as
co-analgesics or adjuvant analgesics as primary analgesics. This
article looks at the assessment and management of pain in the
palliative care setting and the current recommendations for the
management of neuropathic pain.
Pain in the palliative care setting can be caused by the illness
itself, the treatment (radiotherapy, chemotherapy, antiretroviral
treatment), concurrent disorders (tension headaches, urinary tract
infection) or by general debility.1 Thus a very careful assessment and a multimodal approach and understanding of pain are required.
Physiologically the different types of pain can be described as nociceptive (arthritis, cellulitis),2 visceral (pancreatitis, peptic ulcers), neuropathic (herpes zoster, post-stroke pain) and mixed pain, a combination of the other types of pain found in certain disease states such as AIDS and cancer.
Many patients and health care professionals do not
distinguish the pain experience from nociception, which describes the
neural processes involved in the transduction of noxious stimuli.3
Pain in the palliative care setting is not purely a nociceptive or
physical experience but involves all aspects of human functioning:
personality, affect, cognition, behaviour, and social relations.4
Experimental evidence shows that acute persistent pain may promote
biochemical and physiological changes in the peripheral and central
nervous system, which may promote the continuation of pain.5 Chronic pain (lasting longer than 3 months) has a negative impact on quality of life6
and leads to anxiety, depression and sleep disorders. Because pain in
palliative care is multidimensional, a multimodal approach is needed to
address both the physical and the physiological factors (Fig. 1).
Pain assessment
The most important element of pain assessment in
palliative care is to establish a good rapport with the patient and to
show empathy and understanding of his/her pain and suffering. Encourage
the patient to do most of the talking by asking open-ended questions
(Tell me about your pain) and then zone in on more specific questions
(Table I). Specific questions about the location, intensity, character
and temporal profile of the pain, exacerbating factors and concomitant
symptoms will help the clinician distinguish between the different
types of pain.7 Neuropathic
pain is classically described as pricking, tingling, pins and needles,
electric shocks or shooting, hot or burning, numbness, pain evoked by
light touch or pain evoked by change in temperature. This type of pain
is typically confined to a specific set of dermatomes. Nociceptive pain
may be somatic or visceral pain. Somatic pain is typically well
localised and is felt in the superficial cutaneous or deeper
musculoskeletal system. Visceral pain is usually poorly localised,
often described as deep squeezing and/or pressure and may be associated
with nausea, vomiting and diaphoresis, especially when acute. Visceral
pain can be referred to cutaneous sites away of the lesion (shoulder
pain with diaphragmatic lesions).8
The intensity of the pain can be measured by using visual analogue,
drawings and verbal descriptors. A simple tool which is used in
countries with varying degrees of literacy and numeracy is a closed
fist for no pain and each digit indicating an increasing degree of pain
up to five for the maximum. During the assessment the clinician must
also determine how the pain affects the patient’s mood, sleep
pattern, physical activity and relationships. It is also important to
find out what prior treatment has been used and its effect. If possible
it is helpful to interview the family/carer, especially in the
cognitively impaired patients, but not to rely only on proxy reports.
Table I. Clinical assessment of pain2
• Believe the patient’s complaint
• Take a careful history of the pain complaint to place it temporally in the progression of the patient’s illness
•
Assess the characteristics of each pain (site, referral pattern,
aggravating and relieving factors and impact on daily living)
• Clarify the temporal aspects of the pain (acute, chronic, intermittent)
• List and prioritise each pain complaint
• Evaluate response to previous treatment
• Evaluate the psychological state of the patient and the effect of pain on it
• Perform a careful medical and neurological examination
• Order and review appropriate diagnostic test
• Individualise the treatment to the patient’s circumstances and create realistic goals
• Provide continuity of care
• Empower patient and family caregiver to manage the pain, especially breakthrough pain
• Reassess the patient’s pain response to treatment and compliance
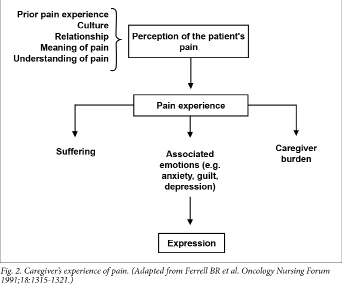
A family member in pain exercises a negative effect
on the dynamics of a family. When patients are facing a
life-threatening illness the meaning and understanding of pain and
suffering must be explored with the family. Family caregivers’
experiences with pain begin with their perception of the
patient’s suffering. This may vary dramatically and is influenced
by many factors (Fig. 2) and may lead to long-term emotional distress
in the caregiver.9
Examination of the patient should start when the
patient walks into the clinician’s office. Look for non-verbal
signs, e.g. the patient who keeps his arm warm or pulls his shirt away
from his body. The physical examination should be to the point and as
non-invasive as possible. In neuropathic pain the clinician should try
to localise the lesion by doing a systemic neurological examination.
This should include a sensory examination to reveal somatosensory
dysfunction, a motor assessment (muscle strength, tonus and fluidity of
movement) and an examination of the tendon reflexes and cranial nerves.
It is also important to assess the peripheral autonomic nervous system
looking at temperature and colour of skin.
Once the clinician has a clear understanding of the
type of pain, its severity, possible cause and emotional component of
the pain, pharmacological and non-pharma-cological interventions can be
implemented. Reversible factors should be addressed and a clear
explanation of the cause of the pain given to both the patient and the
caregiver. Treatment options must be discussed with both the patient
and the caregiver so that they become active participants in the pain
control. It is also important to establish realistic goals with the
patient and to explain that individuals respond differently to
medication and treatments.
Respecting patient autonomy can be very difficult
for clinicians when seeing their patients suffer pain. However, when
patients are facing multiple losses, as most palliative care patients
are, it is very important for the patient to maintain control of
his/her pain management for as long as possible. Patients may be
fearful of using morphine for reasons that need to be sensitively
explored and explained so that patients can take full advantage of
modern pain control methods without significant side-effects.
The World Health Organization has created a
stepwise approach to the management of cancer pain known as the
‘analgesic ladder’ (Fig. 3).10
This analgesic ladder approach is not an evidence-based guideline, but
provides a systemic approach to the management of cancer pain,11 especially in resource-limited areas.
Adjuvant drugs or ‘co-analgesia’ describe any drugs that
have a primary indication other than pain, but are analgesic in some
painful conditions (Table II).13
The diversity and understanding of these drugs has developed over past
decades and some of these drugs are now seen as first-line therapy for
certain kinds of pain.
Table II. Common analgesics for cancer pain
|
Drugs
|
Indications
|
|
Non-steroidal anti-inflammatory drugs
|
Bone pain, soft-tissue infiltration, hepatomegaly
|
|
Corticosteroids
|
Raised intracranial pressure, nerve compression, soft-tissue infiltration, hepatomegaly
|
|
Bisphosphonates
|
Bone pain
|
|
Antidepressants, anticonvulsants
|
Neuropathic pain
|
There is extensive literature regarding the
physiology of the central and peripheral mechanisms of neuropathic pain
and its management. The injury of a neuron produces change in the
excitability in the injured neuron and neighbouring somatic neurons,
causing ectopic action potential discharges. Three factors seem
responsible: up-regulation of voltage gated sodium channels,
down-regulation of potassium channels and possibly a reduction in the
threshold of transient receptor potential (TRP) transducer heat-sensitive channels so that they can be activated at body temperature.13
This ectopic activity may then directly initiate spontaneous sensation
such as burning pain, paraesthesia and dysthethesia.
N-methyl-D-aspartate (NMDA) receptors also play a major role in
hyperalgesia and the enhancement of allodynia, a phenomenon termed
central sensitisation.14
Ketamine, a well-known NMDA receptor antagonist, has been used for many
years in the treatment of neuropathic pain but causes significant
central nervous system side-effects. A major way through which NMDA
receptors work is through the influx of calcium channels and by
decreasing transmitter release with calcium channel blockers (N type)
and α2-δ binding drugs, pain reduction can be achieved.
The International Association for the Study of Pain
(IASP) has developed new treatment guidelines for the management of
neuropathic pain15
guided by improved understanding of the physiology of neuropathic pain
and its management. It is especially in the treatment of neuropathic
pain that new treatment recommendations have to be considered as
first-line therapy. Factors to consider when choosing one neuropathic
drug above another are: pain physiology; side-effects of the drugs;
drug interactions; co-morbidities that can be alleviated by
non-analgesic effect (e.g. depression, anxiety); cost and risk of abuse
and over-dosage. In palliative care time becomes a very important
factor and combination therapy may be needed for rapid onset of pain
relief.
Recommended first-line treatment includes
certain antidepressants (i.e. tricyclic antidepressants (TCA)), through
their action as sodium channel blockers; dual reuptake inhibitors of
serotonin and norepinephrine; calcium channel α2-δ ligands
(i.e. gabapentin and pregabalin) and topical lidocaine. Traditionally
the TCAs have been the most common drug used in the public sector in
South Africa for treatment of neuropathic pain. They are inexpensive
and placebo-controlled studies have provided support for the efficacy
of TCAs in the treatment of neuropathic pain, especially in
post-herpetic neuralgia and painful diabetic peripheral neuropathy.16 TCA in randomised clinical trials have not differed significantly from placebo in the treatment of HIV neuropathy17 and neuropathic cancer pain,18
which are the two most common conditions treated in South African
palliative care. TCAs are cardiotoxic and can cause sedation and have
significant anticholinergic effects.19
Regrettably there is not enough research conducted on the use of
opioids in neuropathic pain despite WHO guidelines that suggest an
analgesic drug in conjunction with an antidepressant or anticonvulsant
for neuropathic pain in oncology patients.
Duloxetine is a selective serotonin norepine-phrine reuptake inhibitor
SSNRI that has shown significantly greater pain relief in randomised
clinical trials compared with placebo in patients with painful diabetic
peripheral neuropathy.20 Its main side-effects are nausea, sedation and dizziness.
Gabapentin and pregabalin are both calcium channel
α2-δ ligands which decrease the release of glutamate,
norepinephrine and substance P. Pregabalin and gabapentin have shown
efficacy in a wide variety of neuropathic conditions and have no
clinically important drug interactions, making them ideal drugs to use
in the palliative care setting. Unfortunately, these drugs are
difficult to obtain in the state sector and palliative care clinicians
would like to see these drugs as part of the essential drug list (EDL).
Gabapentin and pregabalin require dosage reduction in patients with
renal impairment. It is also necessary to start at a lower dosage in
the frail and elderly.
Studies have shown that lidocaine patch 5% improves pain in patients with post-herpetic neuralgia and allodynia,20
but these patches are not available in South Africa. Lidocaine gel was
shown to be effective in pain relief in patients with post-herpetic
neuralgia and allodynia22 but not in patients with HIV neuropathy.23
Opioid analgesics and tramadol are recommended as
third-line treatment in the treatment of neuropathic pain in the
general population. However, in neuropathic pain in cancer, acute
neuropathic pain, episodes of exacerbation of pain and while titrating
up medication these drugs are considered to be first-line medication.
Opioid analgesics have demonstrated effectiveness in reduction of
neuropathic pain.24
Opioid analgesics are the mainstay for the treatment of moderate to
severe pain in palliative care. When administered according to
guidelines, opioids provide adequate pain relief for more than three
quarters of patients with cancer pain.25 The main reason for not prescribing these drugs is still opiophobia among health professionals.26
Mist morphine (liquid morphine) is cost effective, and side-effects
(constipation, nausea, vomiting and drowsiness) can easily be managed.
Respiratory depression is rarely seen in the palliative care setting
where morphine is introduced at low dose, administered orally and
titrated slowly to achieve pain relief. Research has also demonstrated
that the risk of addiction with the medical use of opioids is low.
It is always important to realise that any pain is
not a homogenous entity and requires reassessment and adjustment of
pain medication. The pain a patient has today might not be the pain he
experiences tomorrow because of the physical and physiological changes
patients in a palliative care setting face. Pain control is achievable
in the African setting with good assessment, review and attention to
detail.
Dr Milton Raff for his input and guidance.
References available at www.cmej.org.za
IN A NUTSHELL
• Pain in the palliative care setting can be
caused by the illness itself, the treatment (radiotherapy,
chemotherapy, antiretroviral treatment), concurrent disorders (tension
headaches, urinary tract infection) or by general debility.
• Physiologically the different types of pain
can be described as nociceptive (arthritis, cellulitis), visceral
(pancreatitis, peptic ulcers), neuropathic (herpes zoster, post-stroke
pain) and mixed pain, a combination of the other types of pain found in
certain disease states such as AIDS and cancer.
• Because pain in palliative care is
multi-dimensional, a multimodal approach is needed to address both the
physical and the physiological factors.
• The most important element of a pain
assessment in palliative care is to establish a good rapport with the
patient and to show empathy and understanding of his/her pain and
suffering.
• Once the clinician has a clear understanding
of the type of pain, its severity, possible cause and emotional
component of the pain, pharmacological and non-pharmacological
interventions can be implemented.
• The World Health Organization has created a
stepwise approach to the management of cancer pain known as the
‘analgesic ladder’.
• The International Association for the Study
of Pain (IASP) has developed new treatment guidelines for the
management of neuropathic pain guided by improved understanding of the
physiology of neuropathic pain and its management.




Comments on this article
*Read our policy for posting comments here